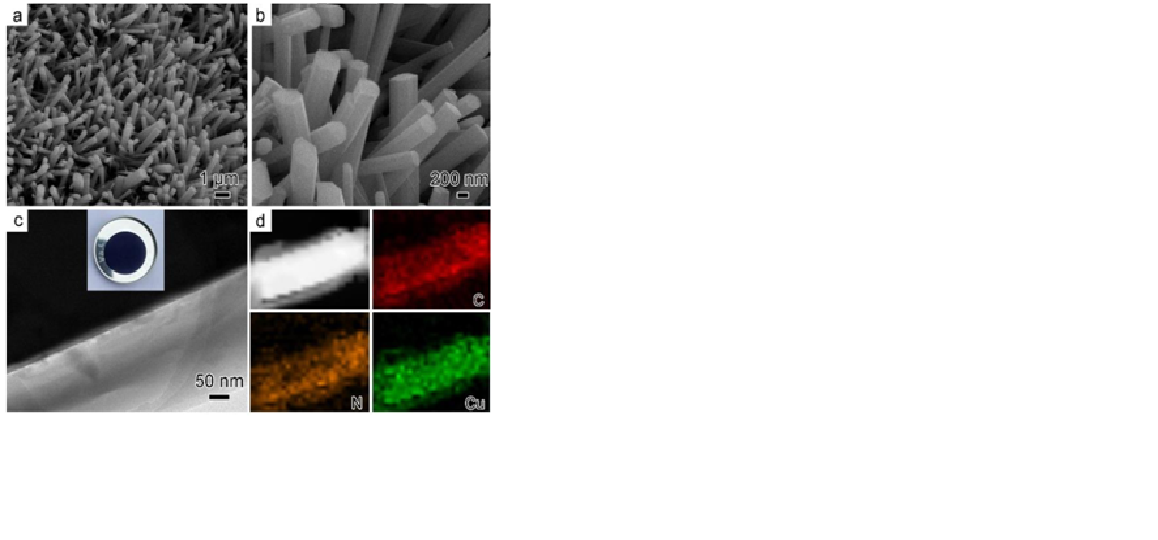
Recently, carboxylate metal‐organic framework (MOF) materials were reported to perform well as anode materials for lithium‐ion batteries (LIBs), despite the alleged lithium storage mechanism of MOFs is controversial. To dig into the mechanism of MOFs as anode materials for LIBs, a self‐supported Cu‐TCNQ (TCNQ: 7,7,8,8‐tetracyanoquinodimethane) film was fabricated via an in situ redox routine, and directly used as electrode for LIBs. The first discharge and charge specific capacities of self‐supported Cu‐TCNQ electrode are 373.4 and 219.4 mAh g‐1, respectively. After 500 cycles, the reversible specific capacity of Cu‐TCNQ reaches 280.9 mAh g ‐1 at a current density of 100 mA g ‐1. Mutually validated data reveal that the high capacity is ascribed to the multiple electrons redox conversion of both metal ions and ligands, as well as the reversible insertion and desertion of Li+ ions into the benzene rings of ligands. This work raises the expectation for MOFs as electrode materials of LIBs by utilizing multiple active sites, and provides new clues for designing ideal electrode materials for LIBs.
本文下载地址:https://doi.org/10.1002/asia.201901190